Co-reporter: Stephen D. Springer, Jian He, Megan Chui, R. Daniel Little, Marcus Foston, and Alison Butler
pp: 3212
Publication Date(Web):April 22, 2016
DOI: 10.1021/acssuschemeng.6b00245
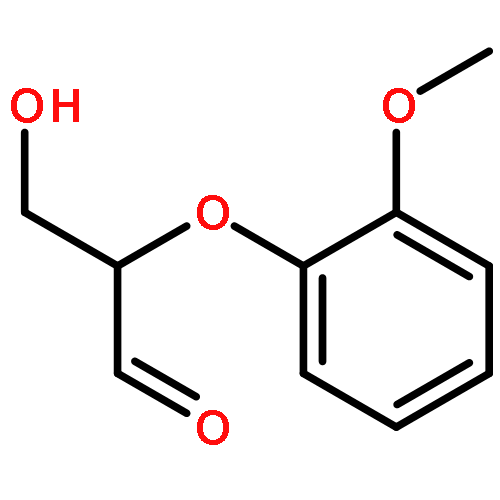
The manganese catalyst, (1R,2R)-(−)-[1,2-cyclohexanediamino-N,N′-bis(3,5-di-t-butylsalicylidene)]manganese(III) chloride, was used to activate H2O2 to oxidize organosolv lignin and a lignin model compound. Oxidation of the β-O-4 lignin model substrate 1-(4-hydroxy-3-methoxyphenyl)-2-(2-methoxyphenoxy)propane-1,3-diol (320.3 m/z) and poplar organosolv lignin resulted in both fragmentation and polymerization processes, likely via phenoxy radical formation. Matrix-assisted laser desorption/ionization (MALDI) time-of-flight (TOF) mass spectrometry (MS) of the reaction products from the β-O-4 model substrate showed oligomers of the substrate with masses of 661.192, 979.355, and 1297.466 m/z that correspond to a dimer, trimer, and tetramer of the β-O-4 model substrate, respectively. Nuclear magnetic resonance (NMR) shows the formation of 5–5 diphenyl and 4-O-5 linkages in the β-O-4 model substrate oxidation products. Gel permeation chromatography (GPC) detected three peaks, corresponding to the β-O-4 model substrate and its oligomers. Products from the Mn-catalyzed oxidation of poplar organosolv lignin by H2O2 were analyzed by GPC, 31P NMR, and 13C NMR. GPC showed an increase by approximately four in the number-average molecular weight of organosolv lignin upon oxidation. NMR shows that polymerization occurs at positions consistent with phenoxy radical coupling, where the observed changes in guaiacyl subunit chemical shifts are most likely due to the formation of 5–5 biphenyl linkages.Keywords: Breakout products; Chemical feedstock; Lignin; Manganese catalyst; Peroxidative oxidation; Polymerization